Leading India towards an Immune Future
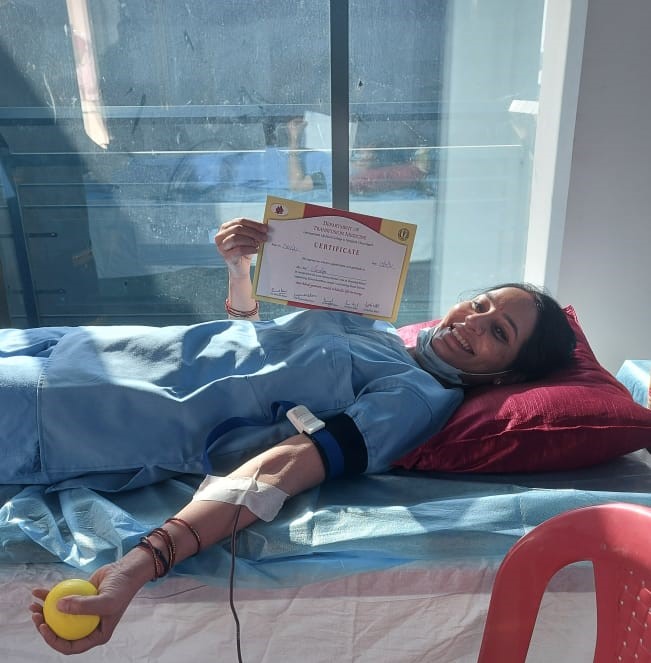
In August 2020, a 53-year-old COVID patient who was in a comatose state, struggling to hold on to life, was infused with Mesenchymal stem-cells taken from an umbilical cord. The results of this infusion were nothing less than enthralling as the patient recovered significantly in over three weeks.
This medical marvel was made possible by Global Institute of Stem Cell Therapy and Research (GIOSTAR), which is on a mission to administer the safest and most effective stem cell therapies and to deliver the highest quality care through its hospitals.
GIOSTAR was founded on the vision to provide stem cell-based therapy to people suffering from all kinds of degenerative or genetic diseases around the world. The company is evidently a leader in developing cutting edge stem cell-based technology, supported by leading scientists with pioneering publications in the area of stem cell biology. GIOSTAR was also one of the first company/research institute in the world which received USA-FDA approval in April 2020 to treat COVID-19 patients with stem cells.
The company’s primary focus is on discovering and developing a cure for human diseases through state-of-the-art and unique stem cell-based therapies and products. The company’s intellectual property, intellectual assets and technology have been developed over its team working on research projects funded to the tune of more than several hundred million dollars.
GIOSTAR is a coalition of esteemed and illustrious scientific minds in the field of genetic and stem cell science. The company’s team members have been involved in the development and utilization of stem cell based clinical protocols related to malignant and non-malignant diseases for the last 20 years.
The company’s leaders are associated with leading universities and research institutes of USA. GIOSTAR is affiliated with University of California San Diego Medical College (UCSD), University of California Irvine Medical College (UCI), Salk Research Institute, San Diego, Burnham Institute for Medical Research, San Diego, University of California Los Angeles Medical College (UCLA), USA, Harvard Medical School, Imperial College, London.
“Our team has developed several research collaborations and has an extensive research experience in the stem cell therapy which is documented by several publications in revered scientific journals,” says Dr Anand Srivastava, the Chairman and Co-founder of GIOSTAR.
Dr Srivastava’s profound success has its root in his unique background and expertise in stem cell biology, protein biochemistry, molecular biology, immunology, in utero transplantation of stem cell, tissue targeting, gene therapy and clinical research.
There are many scientists who can work in a narrowly defined field, but few have broad and multidisciplinary experience to carry out clinical research in a field as challenging as stem cell biology, cancer, and gene therapy. Dr Srivastava’s wide-spectrum expertise is rare in clinical research and perfectly crafted to fit ideally with the GIOSTAR’s projects for stem cell transplant, cancer, and gene therapy research. Dr Srivastava is also serving as stem cell director for the government of Gujarat, India and is in the process of establishing the stem cell program for Gujarat, Government of India.
The team at GIOSTAR is reaching heights of success also under the able leadership of Mr. Deven Patel, the CEO, President and Cofounder. Mr. Patel was honoured with USA Congressional Recognition for his efforts in spreading the advancement of stem cell science around the world. He was also bestowed with the Asian Heritage Award for his business leadership in the field of stem cell science.
GIOSTAR, under the leadership of Mr. Patel, is heading the development of stem cell transplant program for four different State Governments in India with total population of approx. 155 million. The company, in collaborations with Government of Gujarat, India, has founded the world’s largest state-of-the-art stem cell treatment hospital in India. GIOSTAR is also in process of developing additional facilities in China, Thailand, Colombia, Bahamas, Dubai, and Australia.
Talking about how GIOSTAR successfully found the solution to treat COVID-19 related health issues, Dr Srivastava explains, “COVID-19 infection leads to uncontrolled release of proinflammatory cytokines known as ‘cytokine storms’ that potentially lead to fatal damage of many organs of the body. MSCs have specific characteristics that, when infused in the bloodstream, neutralize proinflammatory cytokines (through ‘immunomodulation’), down-regulate the inflammatory gene actions and send messages to regenerate damaged lung and other tissues, which may help in managing COVID-19.” GIOSTAR is also gearing up to start its clinical trial for patients with aftereffects of COVID-19 on lungs and other organs.
We asked Dr Srivastava about his opinion on the necessity for businesses to align their offerings with newer technological developments, especially when it comes to changing industry trends and customer preferences, to which he replied, “Scientific research is an ever-evolving process. GIOSTAR being hard core research organization allows it to be at the forefront of new innovations and possible cure for several incurable disease. This same mindset has made us one of the first research institutes in the world to get very early approval form USA-FDA to treat COVID-19 patients with stem cells.”
Mr. Patel on the other hand shared a few tokens of knowledge through the commercial perspective of regenerative medicine development, he says, “The pharma and scientific research businesses are very competitive, at very high level of science and at very high cost. Therefore, a simple advice to aspiring entrepreneurs is to first get their ducks in the row and get your finances figured out way in advance, otherwise it is very easy to bankrupt the company in this volatile economy.”
GIOSTAR’s plan to continue scaling its operation, keeping in mind the unpredictable future, can be viewed to be agile and ready to run. Pertaining to stable finances, the company’s pipeline of several technologies is very robust and in line with its projections. GIOSTAR is poised to be the largest stem cell research and therapy company on the earth. The company is already becoming a synonym for the stem cell treatment.
Read More : https://insightscare.com/giostar-leading-india-towards-an-immune-future/